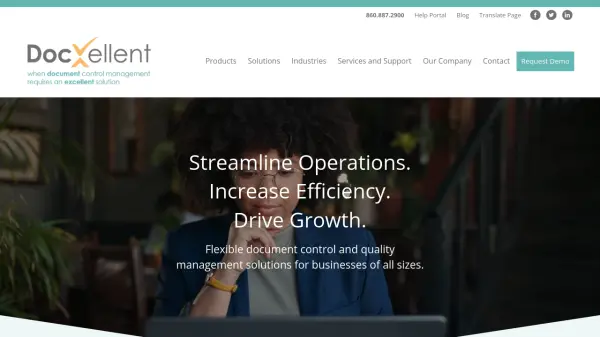
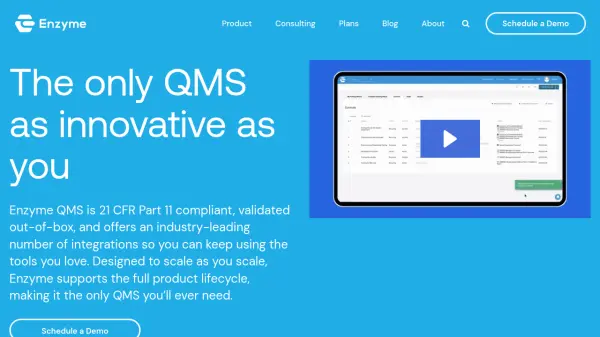
What is ENSUR by DocXellent?
ENSUR from DocXellent provides a comprehensive web-based platform for document management and quality control. The system centralizes critical business documents, securing them in a single repository while enabling automated workflows, revision management, and seamless collaboration among teams. ENSUR supports both validated and non-validated environments, making it suitable for industries with strict compliance and operational requirements.
The software includes features such as customizable electronic forms, version control, training management, CAPA & incident management, and specification management. Users can choose between on-premise and SaaS deployment options, ensuring flexibility and scalability. ENSUR is purpose-built to optimize document-driven processes, align departments, improve data capture accuracy, and maintain consistent standards across various business operations.
Features
- Centralized Document Repository: Securely store and organize all business documents in one platform
- Automated Workflows: Streamline document routing, review, and approval processes with automation
- Version Control: Manage document revisions efficiently and maintain complete audit trails
- Customizable Electronic Forms: Capture and search structured data with configurable forms
- Training Management: Track employee training and manage compliance requirements
- CAPA & Incident Management: Collect, investigate, and resolve corrective and preventive actions
- Specification Management: Seamlessly manage specifications and migrate legacy documents
- Quality Management: Standardize data capture and quality processes across departments
- On-Premise & SaaS Deployment: Choose between flexible hosting options to suit business needs
- Integration Readiness: Supports integration with third-party systems and workflows
Use Cases
- Automating document workflows in regulated industries
- Centralizing and securing confidential business documentation
- Managing employee training records for compliance audits
- Handling version control and approvals for company policies
- Coordinating corrective and preventive action processes
- Migrating legacy content to a digital document management platform
- Streamlining quality management and reporting
- Ensuring compliance with industry regulations such as FDA 21 CFR Part 11 and ISO standards