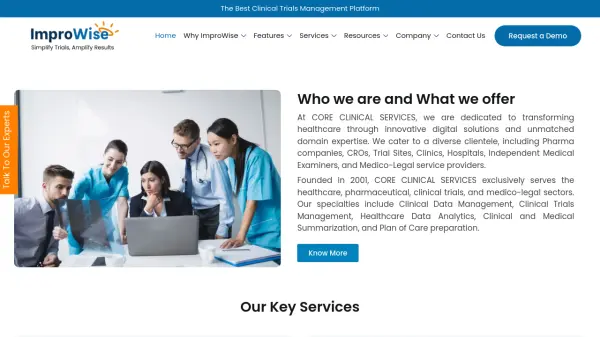
What is Clinical Trials Management?
Clinical Trials Management operates as an Investigative Site Network (ISN) and Clinical Research Organization (CRO) focused on conducting clinical research studies in the Greater New Orleans region. The organization serves as the research arm for Principal Investigators and Sub-investigators, with offices located in Metairie, Covington, Mandeville, and Slidell to facilitate regional research activities.
The company specializes in comprehensive study management and offers additional CRO services including monitoring, auditing, site selection, regulatory compliance, and study start-up support for clients. Their dedicated research team implements the company's vision through expertise in clinical trial execution, contributing to medical advancements through participation in pivotal studies that lead to FDA approvals and publications in prestigious medical journals.
Features
- Investigative Site Network: Conducts clinical research studies across multiple locations in Greater New Orleans
- Clinical Research Organization Services: Provides monitoring, auditing, site selection, and regulatory compliance support
- Study Management: Functions as research arm for Principal Investigators and Sub-investigators
- Regional Coverage: Maintains offices in Metairie, Covington, Mandeville, and Slidell for comprehensive service delivery
Use Cases
- Conducting clinical trials for pharmaceutical companies
- Managing research studies for medical device testing
- Providing CRO services for regulatory compliance
- Facilitating patient recruitment and participation in clinical studies
- Supporting Principal Investigators with study management
Related Queries
Helpful for people in the following professions
Clinical Trials Management Uptime Monitor
Average Uptime
0%
Average Response Time
0 ms